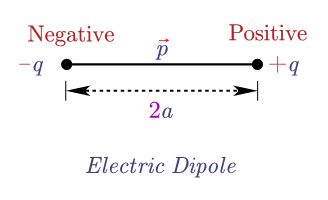
The total dipole moment of a molecule is the vector sum of all the bond dipoles. 'Each end' could mean each end of a bond (each atom), or each end of a molecule, like water. Dipole moment in HCl + and - indicate positive and negative charges, which are separated by distance d. where q is the partial charge on each end and d is the distance between the charges. Polarizabilities and plot them against the relative permittivities. To review, the electric dipole moment is defined as. One can collect the results for dipole moments and isotropic The dipole moment of this system is just p 2QL. This will produce a different output file for every different solvent in the array. A simple dipole is a system consisting of two charges, + Q and Q, separated by a distance 2L. Pam -inp=pcm-prop.inp -replace solvent=$solv -mol=H2O.mol # Now loop over the array elements and substitute them to the string solvent in pcm-prop.inp Solvents are ordered by increasing relative permittivityĭeclare -a solv_array=("N-HEPTANE" "CYCLOHEXANE" "CARBON TETRACHLORIDE" "BENZENE" "TOLUENE" "CHLOROFORM" "CHLOROBENZENE" "ANILINE" "TETRAHYDROFURANE" "METHYLENECHLORIDE" "1,2-DICHLOROETHANE" "ACETONE" "ETHANOL" "METHANOL" "ACETONITRILE" "NITROMETHANE" "DIMETHYLSULFOXIDE" "WATER") The condition for the electronic transition to be allowed is to make the transition moment integral nonzero. where is the electronic ground state and is the electronic excited state. T0 = T : Expectation value zero by time reversal symmetry. The transition moment integral for the electronic transition can be written as. In electromagnetism, the magnetic moment is the magnetic strength and orientation of a magnet or other object that produces a magnetic field, expressed as a vector.Examples of objects that have magnetic moments include loops of electric current (such as electromagnets), permanent magnets, elementary particles (such as electrons), composite particles (such as protons and neutrons), various. S0 = T : Expectation value zero by point group symmetry. *************************** Expectation values ***************************ĭipole length: X : 0.63076018E-12 a.u. Dipole moment of any bond is the product of the net positive or negative charge and distance between the two charged ends, i.e., the bond length.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
